Grand Challenge research harnesses AI to fight breast cancer
Breast cancer has recently overtaken lung cancer to become the most common cancer globally, according to the World Health Organization. Advancing the fight against breast cancer, the BreastPathQ Challenge was launched at SPIE Medical Imaging 2019 to support the development of computer-aided diagnosis for assessing breast cancer pathology.
BreastPathQ Challenge participants were tasked with developing an automated method for analyzing microscopy images of breast tissue and ranking them according to their tumor cell content, to provide a reliable assessment score. As reported in SPIE's Journal of Medical Imaging (JMI), the challenge produced encouraging results that indicate a path toward integrating artificial intelligence (AI) to streamline clinical assessment of breast cancer.
Medical imaging for neoadjuvant treatment
Treatment for large or aggressive breast cancers has often turned to mastectomy as the most reliable therapy. However, therapy known as "neoadjuvant treatment" can result in reduced tumor size, density, and spread, making patients candidates for breast-conserving surgery rather than mastectomy.
Medical imaging allows doctors to assess the effects of neoadjuvant treatment. While the processes of analyzing medical images for cancer detection are typically performed manually and rely on expert interpretation of complex tissue structures, machine-learning algorithms for identifying cancer may increase the reliability and efficiency of those processes. In addition to reducing variability, which is inherent to human pathologists, fully automated methods like these are expected to increase the speed of image analysis.
Intensive focus, international effort
A total of 39 teams from 12 different countries worldwide engaged in the BreastPathQ Challenge. A total of 100 algorithms were developed, validated, and tested. Teams were able to compare their algorithms with those of others from academia, industry, and government, as structured by the Grand Challenge framework, which requires a shared set of source data.
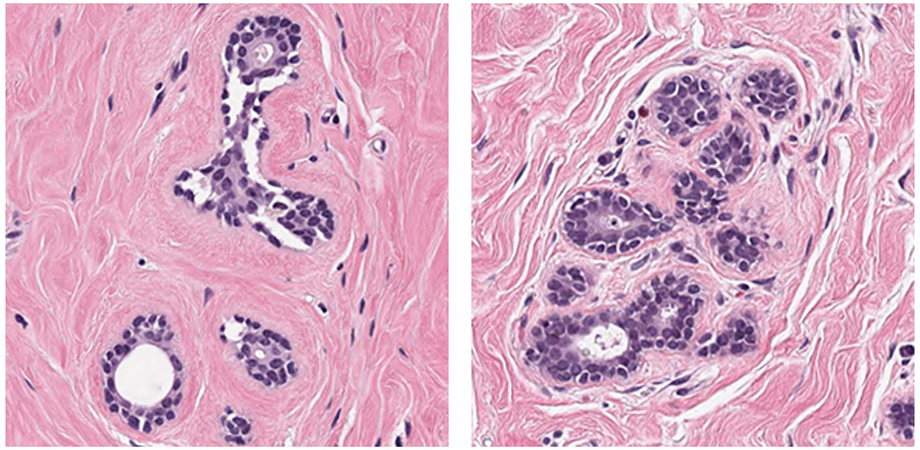
Most of the teams used an ensemble of machine-learning algorithms instead of limiting themselves to a single AI architecture. Top algorithms performed at levels comparable to the pathologists who provided the reference standards for the study, and the best performing algorithm slightly surpassed the scores of the pathologists. The algorithms generally performed well on easier patches of images but struggled on the difficult patches - those for which AI would be especially beneficial to pathologists.

These image patches caused the most difficulty for the submitted algorithms to distinguish healthy cells from cancer. Credit: Petrick et al., doi 10.1117/1.JMI.8.3.034501
The BreastPathQ Challenge was successful because the organizing committee brought together experts in multiple fields. According to Nicholas Petrick, deputy director for the Division of Imaging, Diagnostics and Software Reliability in the US FDA Center for Devices and Radiological Health, and representative for the BreastPathQ Challenge Group, advance collaborative groundwork meant that participants were able to move quickly and efficiently to address the task, access the data set, and develop their algorithms.
Read the open access report by N. Petrick et al., "SPIE-AAPM-NCI BreastPathQ Challenge: an image analysis challenge for quantitative tumor cellularity assessment in breast cancer histology images following neoadjuvant treatment," J. Med. Imag. 8(3), 034501 (2021), doi 10.1117/1.JMI.8.3.034501.
| Enjoy this article? Get similar news in your inbox |
|



